Enhancements in NBS 1.1.7
As of January 29, 2008, release 1.1.7 of the NEDSS Base System is running in our production environment. This new version includes many new enhancements which will improve performance and ease of use as well as providing new functionality to NBS users. The most notable enhancements are listed below.
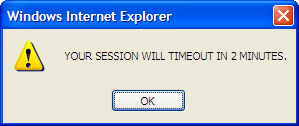
A session timeout clock was added that allows the user to see the amount of time that remains before their session will timeout. The timeout clock appears on the bottom left corner of the browser window. The timeout clock is reset each time the user navigates to a new page in the NBS. When there are two minutes left before the session timeout, the following pop-up window will appear on the page.
Change name of “My Program Area’s Investigations” queue to “My Program Area’s Open Investigations”: The “My Program Area’s Investigations” queue was renamed to “My Program Area’s Open Investigations” to clarify that only investigations with an Investigation Status equal to “Open” are included in this queue.
Patient, Provider and Organization Search: Change Default to “Starts With”: It was discovered during performance testing that using the “Starts With” operator when searching for a Patient, Provider and Organization resulted in faster search results. As a result of this finding, the default operator was changed from “Contains” to “Starts With”.
The following fields were added to the Mumps Investigation Form (Epidemiologic Section) at the request of the Mumps program:
- Did parotitis or other mumps-associated complication onset occur within 12–25 days of entering the USA, following any travel or living outside USA? (Import Status) (MUM136)
- If this is a U.S.-acquired case, how should the case be classified by source? (MUM137)
Foodborne Investigation Form Changes: The following changes were made at the request of the FoodNet program:
Lab Report
- Added the following values to the Resulted Test drop-down:
- E. coli shiga toxin test, EIA or immunoassay
- E. coli shiga toxin test, PCR
- Added the following values to the Coded Result drop-down
- Shiga toxin 1 positive
- Shiga toxin 2 positive
- Shiga toxins 1 & 2 positive
- Undifferentiated
The following changes were made at the request of the National Immunization Program:
Tetanus
- Added “Home” and “Auto” to the code set associated with the question, “What was the environment where acute wound or injury occurred?”
- Added “Stellate Laceration” and “Linear Laceration” and changed “Laceration” to “Laceration Unspecified” to the code set associated with the question, “Principle acute wound or injury type”
- Added “Convalescing” to the code set associated with the question, “Final outcome”
Measles
- Changed the label of the question “Is this case traceable (linked) to an international case? to “Is this case traceable (linked) to an international import?”
Rubella
- Changed the label of the question “Is this case traceable (linked) to an international case?” to “Is this case traceable (linked) to an international import?”
Please let us know if you encounter any issues with the new version or if you have any suggestions for future improvements to the system.